All of us have constantly heard about food allergies, and in fact, 8% of the world population suffers from some type of food allergy. Food allergies can really get in the way of normal life, forcing some to abstain from normal delights most of us take for granted. Those who suffer from this ailment have to live in a state of constant awareness, from having to avoid cheese to prevent uncomfortable symptoms or being on the watch for peanuts to escape an unwanted emergency room visit. But what causes food allergies?
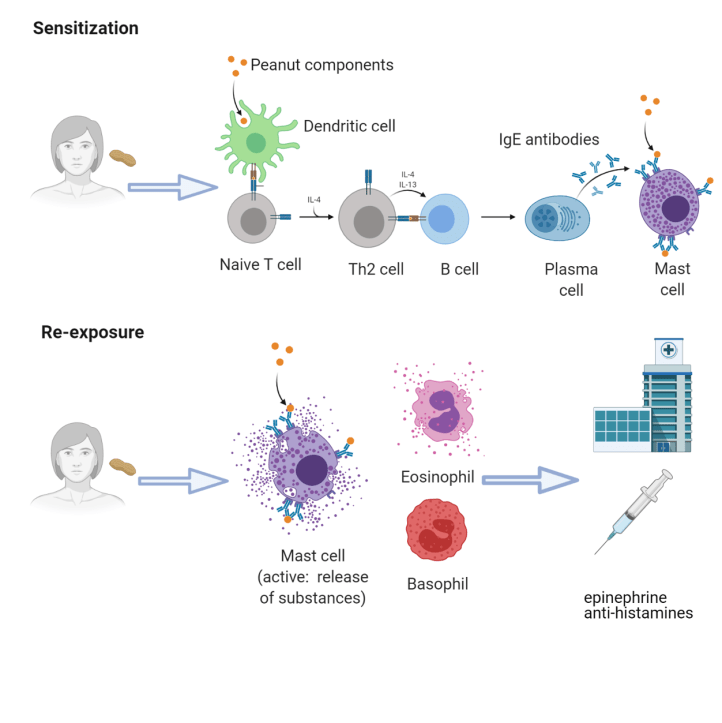
Allergies in general are disorders of our immune system caused by a hypersensitivity reaction. Our immune cells can sometimes “overreact” to normally harmless substances, such as pollen, peanuts, or cat dander, which are referred to as allergens. Allergic reactions are caused specifically by the production of the antibody immunoglobulin E (IgE), which are proteins that normally help to neutralize bacteria or viruses and activate other immune cells. IgE production in B cells is induced by a subset of the CD4+ helper T lymphocytes known as Th2 cells. Th2 cells produce and release interleukins IL-4, IL-5 and IL-13 that can maintain an environment in which further development of Th2 cells is favored. Interleukins are a type of cytokine, or messenger molecules that mediate the communication of immune cells with other cells in the body. IL-4, IL-5 and IL-13 normally help coordinate an immune response for the clearance of parasites or other such extracellular pathogens, but in this case exacerbate allergic reactions. Th2 cells produce IL-4 through the activation of the transcription factor STAT6, which is a protein that can bind to DNA and promote the synthesis of IL-4 and other important immune-modulating proteins. During allergic reactions, Th2 cells stimulate B cell production of IgE, which in turn activates basophils, eosinophils and mast cells. These are types of immune cells filled with different substances in repositories, known as granules, which when released, induce inflammation and cause the symptoms more commonly associated with an allergic reaction.
People who suffer from food allergies develop strong IL-4-producing Th2 responses and IgE production following their first encounter with certain components found in food, such as in the case of nuts, milk products, or seafood. This process is called sensitization (Figure 1). If the person comes into contact with the same food allergen again, their sensitized mast cells will release their granules, leading to a strong allergic reaction.
Regulatory T cells, guardians of the gut
One key immune cell that helps to regulate immune responses are the aptly-named regulatory T cells, or Tregs. Tregs are another subset of CD4+ T cells that have the ability to inhibit the function of other immune cells in their environment and can protect us from over-active immunity, collateral tissue damage, and autoimmunity, allowing us to maintain self-tolerance. Self-tolerance is crucial in allowing our immune system to recognize our cells as ‘self’ as opposed to ‘foreign’ bodies that need to be targeted and destroyed. Studies have shown that Treg function is perturbed in some instances of allergies and autoimmune diseases.
Tolerance to food (oral tolerance) is dependent on Treg development from CD4+ T cells in the presence of TGFβ1. TGFβ1 is a well-known immunomodulatory cytokine, helping to control immune responses. It can inhibit the proliferation of macrophages, regulate different subsets of T cells, control the activation of neutrophils and endothelial cells and promote tissue repair after local immune responses. TGFβ1 is produced not only by Tregs, but also by monocytes, macrophages, and other T cells. However, the exact role of TGFβ1 produced by Tregs in the development of oral tolerance is not fully understood. Turner and colleagues from Boston Children’s hospital wanted to dig in and understand more. They demonstrated that the TGFβ1 derived specifically from Tregs is essential in controlling allergic and autoimmune responses in the gut, and it is very finely tuned by the number of copies of the Tgfb1 gene and the presence of microbiota in the gut.
When is tolerance to food lost?
In this study, the research group developed a genetically-modified mouse model of food allergy, which was achieved by inducing a state of hyperactive IL-4 signaling. With this excess of IL-4, they found that Tregs derived from these mice behave less like Tregs and more like Th2 cells, which are involved in allergic reactions. On the other hand, they noticed that if they eliminated the molecules IL-4, STAT6 and IL-13 from Tregs, the change of behavior from Treg to Th2 cells was not seen anymore. These interesting results suggest that IL-4 signaling is deeply connected to the behavior of Tregs in the gut, and could potentially be linked to food allergy development in these mice. To further understand what is happening inside these Th2-like Tregs, the researchers wanted to explore how IL-4 was impacting Treg function at the molecular level.
Another gene expression control mechanism they studied was the epigenome. The epigenome is a mix of chemical compounds and proteins that directly attach to and interact with DNA to control the production of proteins, essentially turning genes on or off. Turner et al. looked at the epigenome of Tregs in their allergic mice and found that the Tgfb1 gene was not very active. When they genetically removed the transcription factor STAT6 from all the cells, however, this was reversed. This means that the Tregs in allergic mice, which behave more like Th2 cells, suppress the Tgfb1 gene transcription through an essential molecule in IL-4 signaling, STAT6. With all these experiments, they demonstrated that the Th2-like behavior of Tregs in allergic mice is associated with decreased TGFβ1 production by Tregs and no other cellular sources, leading to the development of food allergy.
Tregs need both copies of the Tgfb1 gene
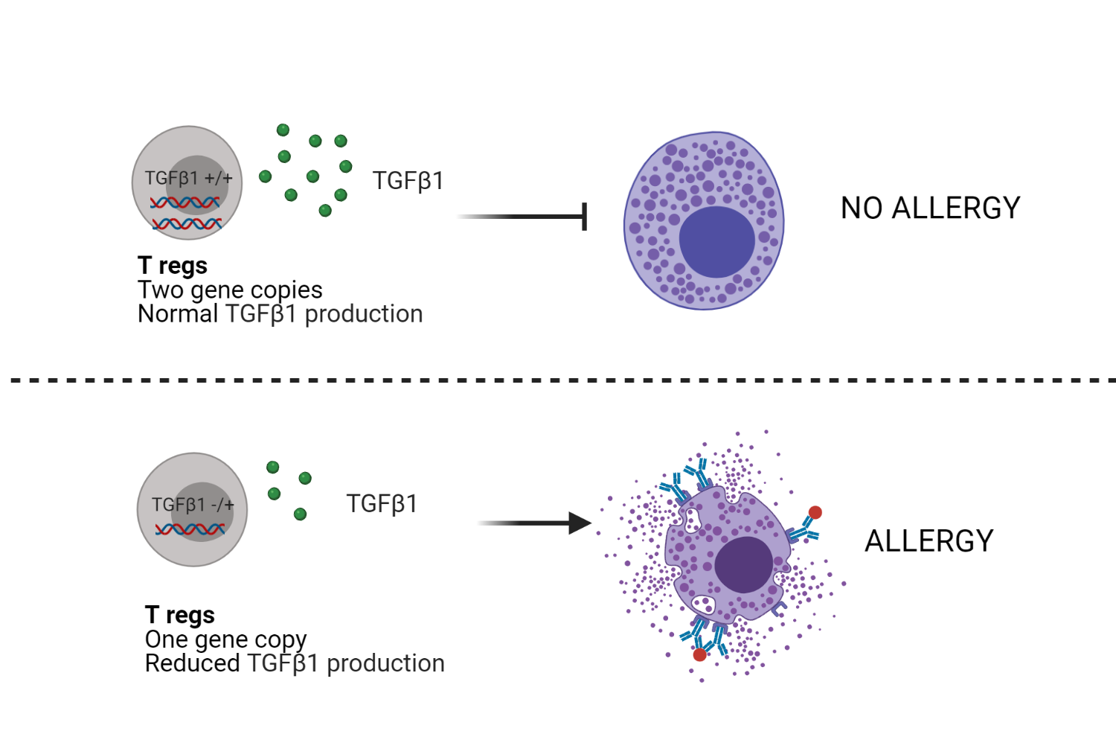
Turner and his team of researchers wanted to further investigate what the impact of decreased TGFβ1 production in Tregs has on food allergy. To do so, they made a new mouse model, in which Tregs carried only one copy of Tgfb1 gene, instead of the two normally inherited by both parents. Additionally, these mice were made susceptible to a common protein known as ovalbumin (or OVA, a protein derived from chicken egg), that is a strong allergen widely used in research. When these sensitized mice were exposed to OVA again, they developed anaphylaxis, which is a severe form of immediate hypersensitivity, characterized by a lack of breath, tongue swelling, rash, and low blood pressure, like the reaction some people suffer from if they have a strong peanut allergy. As expected, these mice also showed high concentrations of IgE, increased Th2 responses, and highly active mast cells when compared to mice that carry both Tgfb1 gene copies. These results confirm that the total TGFβ1 produced specifically by Tregs is essential for controlling allergic reactions, which is tightly linked to the expression of both Tgfb1 gene copies in these cells (Figure 2).
Microbiota as an ally against allergies
Microbiota, or commensal bacteria, are microorganisms normally present in diverse places of our body such as the gut, mouth, nose, and skin. These microorganisms have been shown to have important effects on our immune system.
For example, previous studies have described a role for microbiota in the differentiation of Tregs in the gut. Some well-studied microbiota include the Clostridiales and Bacteroidales species, which live in the gut and help inhibit the development of certain gut pathologies, including food allergy. Turner et al. saw that the presence of microbiota is essential for Tregs in the gut to produce TGFβ1. When they treated mice susceptible to OVA that also lacked a copy of Tgfb1 gene with Clostridiales bacteria, these mice did not develop anaphylaxis as seen in their previous experiments, when the bacteria was not present. It seems, therefore, that the microbiota helps to prevent the development of food allergies by inducing the production of TGFβ1 in Tregs , which is crucial for the regulation of immune responses in the gut.
In summary, this group of scientists identified that Tregs are the essential source of TGFβ1 important in preventing immune responses to common food allergens, therefore avoiding food allergy and autoimmunity. It is not only important that TGFβ1 is produced, but also that it is produced specifically by Tregs and that both copies of the gene are active, which means sufficient TGFβ1 is produced. Although more research needs to be done, this study opens the possibility for the use of Tregs in cell therapy for treatment of allergies and autoimmune diseases by modifying their production of TGFβ1. In addition, the use of probiotics, specifically focused on the Clostridiales species, could benefit Treg function and improve oral tolerance. Maybe in the near future, we can dream of happily eating all the food that, at least for now, can earn us an unwanted trip to the emergency room.
_________
This article summarizes work from Turner and colleagues:
Turner JA, et al. Regulatory T Cell-Derived TGF-β1 Controls Multiple Checkpoints Governing Allergy and Autoimmunity. Immunity. 2020 Dec 15;53(6):1202-1214.e6. doi: 10.1016/j.immuni.2020.10.002.