One of the most puzzling medical mysteries, which still remains a leading cause of mortality in hospitals today, is sepsis. Imagine going to the hospital for say, kidney stones, or to treat a sudden injury. Nothing too life-threatening right? But hospitals happen to be inundated with sick people, and sick people tend to bring all kinds of germs with them. Your simple trip to the hospital may end up getting a lot more complicated if you happen to catch one of these bugs. An innocuous infection can end up becoming a life-threatening condition if sepsis were to develop, and your hospital stay may suddenly be a lot longer than expected.
Sepsis, a long shadow in our medical history
The medical definition of sepsis has changed many times over the years, and its latest definition was updated as recently as 2016 as “a life-threatening organ dysfunction caused by a dysregulated host response to infection.” In simple terms, sepsis is the point at which the force of our own immune response to infection turns against us and overwhelms our body, leading to the shutdown of organs and eventually, death. Sepsis is usually observed when an infectious agent, specifically bacteria, reaches the bloodstream and is able to freely travel through the body, initiating an overwhelming host response. However, the mechanisms that initiate this immune storm are not well understood, and treatment is therefore lacking. The best we can do now is to identify the clinical symptoms as early as possible and try to mitigate organ damage while also combating the infection with antibiotics. Even if a patient survives, the long-term outcome may be gloomy, with the possibility of physical and cognitive damage that cannot be undone.
In October of last year, Borong Zhou and a group of researchers published an article in the journal Nature Microbiology that may provide a feasible target for treatment of sepsis. This exciting work examined the role of a protein known as sequestosome-1 (or p62) in the induction of an inflammatory state in immune cells during sepsis both in cell culture and in various mouse models of sepsis. Furthermore, samples derived from sepsis patients showed a positive correlation between the presence of p62 in patient samples and poor clinical outcomes, suggesting that p62 could become a possible target for treatment of sepsis in humans. Although this type of research is at very early stages, it does bring hope in increasing our understanding of sepsis, which has continued to plague us since time immemorial.
The release of p62
In this recent study, researchers examined the role of the protein p62 in modulating immunity during sepsis. Specifically, they focused on the release of p62 from an important group of immune cells, monocytes and macrophages, which are one the first and most numerous responders to infection that lead to inflammation. They were able to identify two unique models of p62 release, labeled as either active release or passive release. In the active model, they showed that following monocyte/macrophage stimulation with LPS (a bacteria molecule), there was accumulation of extracellular p62 outside of the cell over 12 hours. The passive model involved another mechanism of p62 release in which the cells are instructed to burst and release many molecules into their environment, including p62. Importantly, the release of p62 can shape the immune response of neighboring cells. After establishing a consistent release of p62 from monocytes and macrophages following stimulation, the next question was to identify the impact of p62 release on surrounding cells, and how it can shape immune responses during sepsis.
How does p62 induce inflammation?
One of the interesting clinical diagnostic markers of septic shock, a more serious form of sepsis, is increased levels of lactate in the serum, a condition known as hyperlactatemia. Lactate is the metabolic output of glycolysis, or the breakdown of sugar in the cell to produce energy. There is a very close link between increased glycolysis and inflammation, and most immune cells, including macrophages, tend to become more inflammatory when they have increased levels of glycolysis. Based on this observation, the researchers investigated whether the release of p62 was involved in increasing the glycolytic rate of macrophages and found that this was the case.
Further analysis showed that with this increased glycolysis, there was a change in the macrophages toward a more inflammatory state, as expected. Macrophages, which need to respond to a variety of complex stimuli in the body, need to maintain a certain flexibility in order to respond appropriately in different situations. Therefore, depending on the type of response that is needed, these cells can either become a more inflammatory type of cell, known as M1, or an anti-inflammatory type, known as M2. The M1 phenotype usually develops under conditions of infection, which allows them to better kill and clear out bacteria. However, too much inflammation can lead to unintended tissue damage, which is a hallmark of sepsis. In this study, scientists showed that increased glycolysis mediated by p62 was leading to M1 polarization, as they could see increased expression of hallmark inflammatory markers in these cells after 24 hours of stimulation.
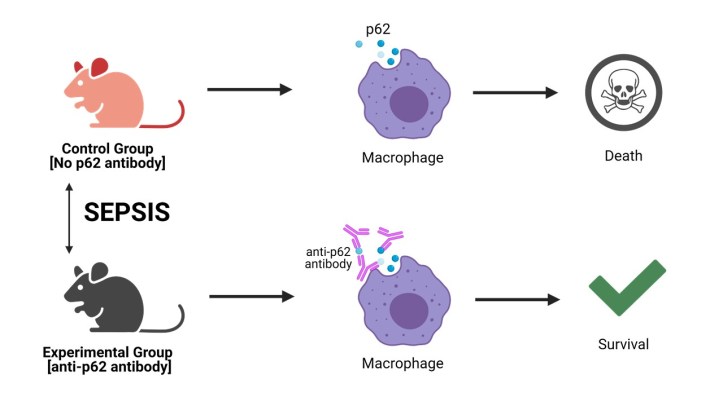
Figure 1: Treatment of mice with anti-p62 antibody. Sepsis was induced in two groups of mice. The control group received no treatment and the experimental group received an anti-p62 antibody injection. This antibody binds to the released p62 and blocks its function. Survival was then tested over time. Figure created with Biorender.com
Of mice and men: p62 has an important role in sepsis
Defining the signaling pathway by which p62 can directly influence the outcome of disease is fascinating, but of course remains meaningless until proven to be relevant inside living organisms. To accomplish this, the research group tested the outcome of sepsis in various groups of mice in which they could control the activity of p62. These models showed that treating mice with an antibody, or blocking molecule, that specifically neutralizes p62, leads to significantly increased survival compared to groups that did not receive the treatment. These experiments corroborated their previous findings that p62 is directly mediating the inflammatory effects of sepsis.
Interestingly, they were also able to show a connection between p62 and sepsis in humans. By comparing blood samples from patients that survived sepsis versus those that did not, they found a positive correlation between p62 levels and expression of inflammatory modulators that can lead to organ damage. Together, these observations form a strong foundation to suggest that p62 indeed has a role in exacerbating an inflammatory response in monocytes and macrophages during sepsis. This is, at least, the first step toward unraveling sepsis at a molecular level, as well as opening new avenues of research for treatment of sepsis, which we have thus far sorely lacked.
This article summarizes work by Borong Zhou and colleagues:
Zhou, Borong, et al. Extracellular SQSTM1 mediates bacterial septic death in mice through insulin receptor signalling. Nature microbiology 5.12 (2020): 1576-1587.
Cover image courtesy of Pexels images.