
In humans and other vertebrates, the innate immune system represents the “first line of defense”, which recognizes potentially dangerous molecules that threaten the functioning equilibrium of the body. Environmental damages caused by chemicals, radiation or microbial infections in the living cell need to be detected and identified to launch appropriate removal, repair and recovery responses. To this end, the body has genetically evolved to discriminate molecules as self or non-self/foreign as well as benign or dangerous.
Interestingly, one such danger molecule recognized by the innate immune system is DNA itself. DNA is the hereditary material in multiple organisms including many viruses, bacteria and humans, but within a host cell, it is exclusively enclosed in the nucleus, wrapped in a highly condensed structure called the chromatin. Hence, the cell recognizes DNA present outside of the nucleus, i.e. in the cytoplasm, as an irregular behavior. If the host cell detects DNA outside the nucleus, it considers the DNA as a material of microbial origin or as a derivative of its own damaged chromatin, and initiates proper immune responses. Scientists have recently made new discoveries about the function of an immune “sensor” called cGAS (or cGAMP synthase) which activates the immune system to combat the threat of “rogue” DNA. Further knowledge about cGAS can provide insights into how the host immune system differentiates between harmful and innocuous DNA.
A critical step in understanding the function of a protein is to know where it is localized. Considering that cGAS was discovered as an activator of cytosolic DNA, it was largely assumed that its inactive form was present in the cytoplasm. However, studies have observed that cytosolic cGAS only represents a very small fraction of the total protein found in the cell. A consensus about inactive cGAS localization, and its activation upon DNA recognition is yet to be reached—and it appears that the answer is not so straightforward.
Research from independent groups observed that besides being in the cytoplasm, cGAS could also be present in the nucleus or tethered to the plasma membrane (the lipid-bilayer membrane enclosing a cell) prior to activation, poised to respond appropriately to foreign or self-derived DNA fragments.
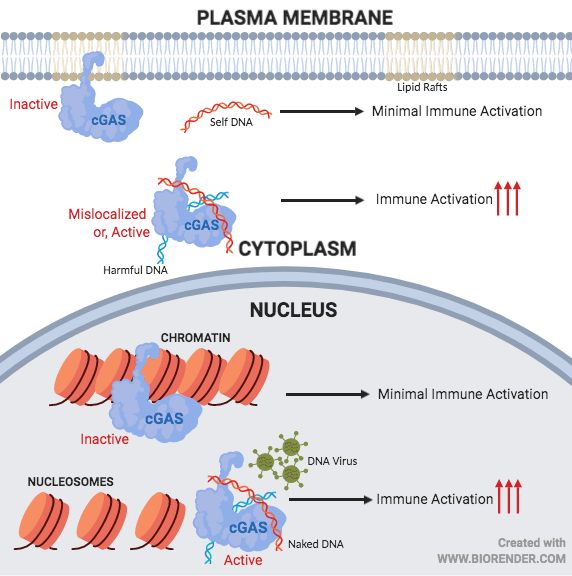
Barnett et al. observed that cGAS was enriched in the sub-domain of plasma membrane known as lipid rafts (which contain high concentrations of molecules like cholesterol and glycosphingolipids). cGAS was secured to the membrane by the interactions between its positively-charged, DNA interacting N-terminal and certain negatively-charged lipids in the membrane. This tethering prevented cGAS from being activated by the harmless, endogenous DNA. The researchers also observed that a mutant cGAS lacking its membrane-binding N-terminal domain localized both in the cytosol and nucleus. The mutant cGAS also exhibited an increased immune response in an unaffected cell as well as in a stressed cell (e.g. chromatin damage induced by chemicals that generate cytoplasmic DNA fragments) indicating that cGAS localization is critical in limiting its exposure to self-DNA.
cGAS is important for antiviral responses, but the majority of DNA viruses have an exclusively nuclear life cycle—in other words, viruses infect cells, live and replicate within the nucleus. Current hypotheses predicting cytoplasmic localization and activation of cGAS rely on the theory that viral DNA “leaks” into the cytoplasm during cellular entry or exit.
The idea that cGAS is primarily located in the nucleus warrants the obvious questions: how is cGAS inactive in the presence of abundant chromatin DNA, and how does cGAS get preferentially activated during DNA damage or viral infection?
Following independent studies in tumor models that observed cGAS localization in the nucleus, Gentili et al. attempted to understand the DNA-sensing function, if any, inside the nucleus. Consistent with Barnett et al., they show that the short N-terminal fragment remained in the nucleus. However, a longer N-terminal domain was cytoplasmic revealing that it contained the motif responsible for keeping cGAS outside the nucleus. They also reported that nuclear cGAS displayed higher activity in resting cells, but initiated a stronger immune response upon DNA transfection.
In striking contrast to these studies, Volkman et al. found that majority of cGAS was in the nucleus irrespective of whether cells are actively dividing or not. They observed that endogenous, inactive cGAS present primarily in the nucleus could be activated to produce cGAMP upon DNA transfection. They show that endogenous cGAS is tightly bound to the nucleus that requires an intact, undamaged chromatin. Similar to Barnett et al. and Gentili et al., they also observed distribution of the small N-terminal cGAS fragment in both nucleus and cytoplasm, but the longer mutant lacking the interacting domain was retained completely in the nucleus of the cells.
So, how will nuclear cGAS differentiate between damaged DNA and the healthy chromatin? Volkman et al. suggests a model of regulated untethering of the cGAS. It suggests that cGAS is bound indirectly to chromatin DNA. Upon detecting damaging foreign or self-DNA in the nucleus, cGAS will be released from the chromatin and accumulate on the nucleic acid fragments to initiate immune signaling.
Of course, these observations raise new questions about how the optimum activation state of cGAS is reached in the presence of damaging DNA? What are the factors that assist in switching between an inactive and active cGAS in the nucleus?
A study of cGAS during cell division by Zeirhut et al. shows that nuclear cGAS is bound to the DNA/protein complexes within the chromatin called nucleosomes. Interestingly, binding of cGAS to nucleosomes is much stronger than cGAS bound to naked DNA. However, this tight-binding to nucleosomes impedes cGAS activation, which signals more effectively when bound to free DNA. Meanwhile, Lahaye et al. discovered the requirement of a host protein during HIV infection that recognized infecting viral particles and mediated virus recognition by cGAS in the nucleus.
Being a central molecule in detecting damaging DNA molecules, it is not surprising that cGAS is ubiquitous, and present in all the cell compartments where it has the highest probability of coming across its target molecules. Spatial distribution of cGAS within the cell is critical because improper activation of cGAS would be responsible for incorrect immune response, that would promote autoimmune activation leading to disease. Understanding the mystery of cGAS localization, and its regulation, will help elucidate how the innate immune system has evolved to discriminate between self and non-self, as well as in detecting harmful self-derived danger molecules.
Featured Image via bionicteaching and licensed under CC BY-NC 2.0