Imagine if doctors could harness the power of your own immune system to target cancer cells like a homing missile. Our immune system packs a punch when fighting off diseases, but due to the cloaking mechanisms used by cancer cells, it can miss the mark.
Chimeric Antigen Receptors (CARs) can give our immune system the power to see dangerous cancer cells in a whole new way. CAR technology is at the intersection of genetic engineering, immunology and personalized medicine. CARs are specifically engineered molecules that can be inserted into our own immune cells to identify markers only expressed in cancer cells. These receptors are already being used to treat patients with certain types of cancers that come from the blood like leukemia and lymphoma. However, this approach has been largely unsuccessful in treating other types of tumors, at least until now. The development of CARs for macrophages, a type of immune cell that can infiltrate tumors, could be the next step in the treatment of solid tumors like breast cancer and possibly many others.
Cancer cells hide from the immune system
Although our immune system can efficiently recognize and protect us from most pathogens and dangerous cell mutations, sometimes it needs a little extra help. When cancer develops and starts to take on dangerous characteristics, it needs to cloak itself to continue to grow inside the body without alerting our immune system.
Our immune system is composed primarily of two arms, the innate and the adaptive immune system. The innate immune response is a broad first line of defense, while the adaptive immune response is more targeted toward eliminating a specific threat. T cells are an important part of the adaptive immune system that can recognize very unique molecular patterns expressed by pathogens and other threats, like cancer cells. Once these T cells are engaged, they can orchestrate a targeted immune response, clearing the threat and generating long-lived memory cells that can provide life-long protection.
The molecule on T cells responsible for recognizing these unique molecular patterns is known as the T cell receptor (TCR), which is expressed on the surface of these cells and can interact with other cells in their environment. Our adaptive immune system contains a vast repertoire of these T cells in order to increase our chances of recognizing foreign molecules that may cause us harm. Cancer cells, however, can avoid detection by both the innate and adaptive immune system, making our defenses blind to the growing threat.
CARs give T cells the ability to target cancer cells
CAR T cell therapy works by engineering components of the TCR to make a new molecule that has everything needed to recognize and activate the T cell to eliminate a target, in this case cancer cells. Instead of waiting for the immune system to find the dangerous cell by random chance, the recognition part of the engineered receptor can be programmed to target a specific marker expressed only by cancer cells.
A patient’s T cells are collected from a blood sample, and then the DNA that encodes the CAR is added to the isolated cells. The T cells now expressing this CAR are infused back into the patient where they direct the immune system to attack and kill the cancer cells. There are currently two forms of CAR T cell therapies that are FDA-Approved to treat patients and outcomes are promising. This has been a huge progress for treating Acute Lymphoblastic Leukemia (ALL), the most common blood cancer in children. Before the approval of CAR T cell therapy, there were no effective treatments after relapse. Despite early successes with this treatment method, one of the challenges has been adapting it to other kinds of cancers. Several factors that differ in solid tumors make this T cell strategy difficult to use. That’s where macrophages, an important component of the innate immune response, come into the picture.
CAR-Macrophages open doors to treating solid tumors
Macrophages are a white blood cell of the innate immune system, which means that they are not naturally able to “learn” or generate long-term protection the way that the adaptive immune system does. Macrophages are able to engulf and ‘eat’ pathogens, dying cells, or even cancer cells, but they only recognize very broad danger signals and are easily tricked by cancer. Since solid tumors are anchored to one place in the body, they have to manipulate the surrounding cells to survive. Cancer cells hijack the surrounding tissue and take advantage of non-cancerous cells to promote the generation of cell repair signals that your body would normally use to fix itself after an injury or illness and use them to thrive and grow in what is referred to as a tumor microenvironment. Macrophages play a role in promoting this environment if they are not able to recognize the cells as cancerous. However, scientists are now trying to take advantage of macrophages’ ability to get close to the tumor by using CARs to reprogram them to see the tumor as dangerous.
The components that make up the CAR come from a T cell receptor, but scientists demonstrated that macrophages can use these same signals to cause a response that leads to engulfment and digestion of the targeted cancer cells. The T cell receptor component called CD3ζ (Figure 1A) shares similarity with the macrophage component that signals macrophages to start eating. One major challenge that researchers had to overcome was getting the DNA that encodes the CAR inside of the macrophages. Macrophages are naturally resistant to genetic changes because they need to protect themselves from viruses and other pathogens. To overcome this hurdle, a team of researchers at the University of Pennsylvania School of Medicine used a special virus called Ad5f35 that can dock onto macrophages and deliver the DNA effectively (Figure 1B). The teams successfully showed that these CAR macrophages could shrink tumors and increase survival time in mice (Figure 1B). Phase 1 clinical trials have begun to apply this CAR macrophage strategy to HER2 overexpressing cancers, and the first patient has been dosed as of March 2021.
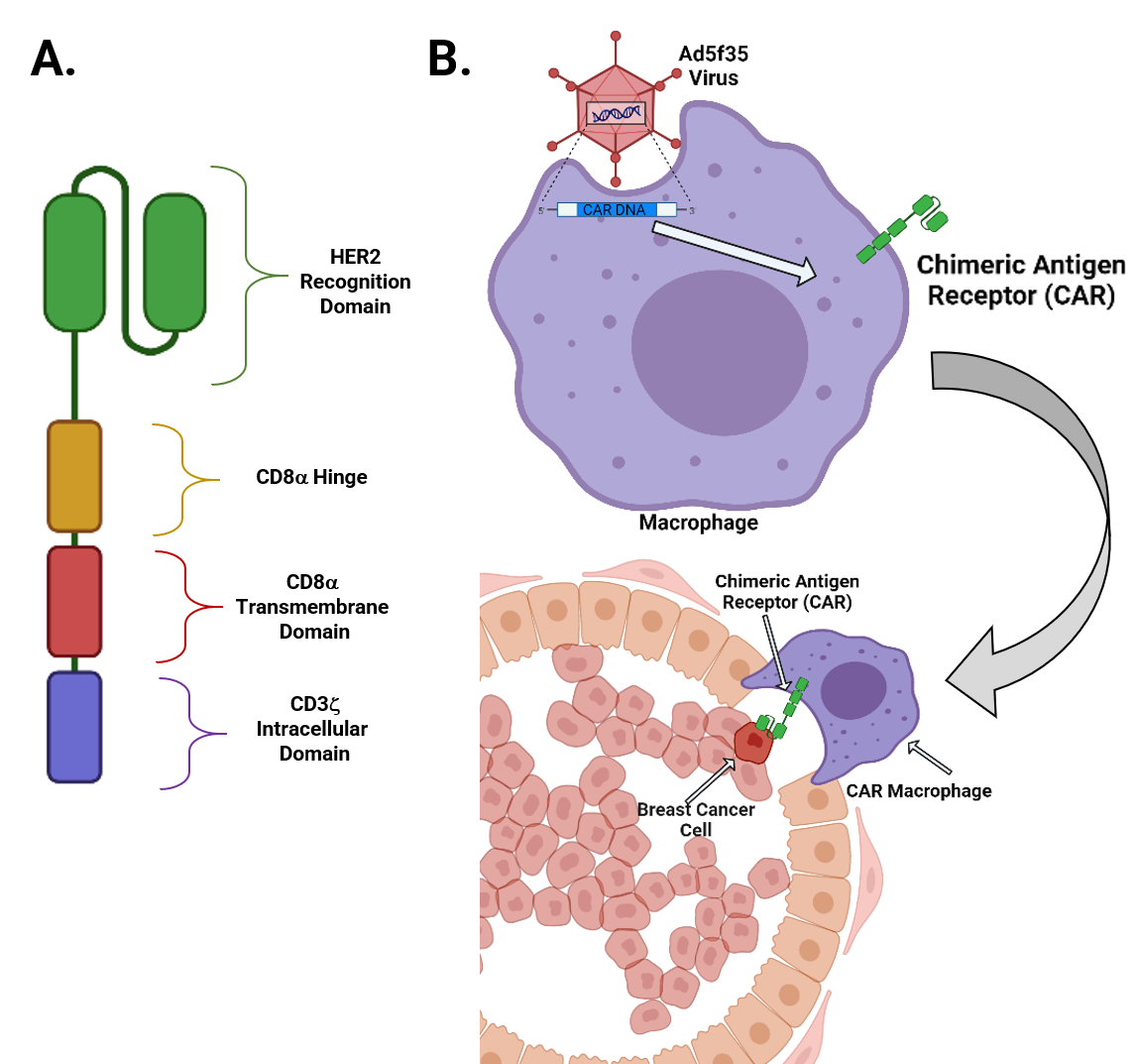
Figure 1: Making a CAR-Macrophage to Treat Solid Tumors. A) The Chimeric Antigen Receptor (CAR) engineered by Klichinsky et al. was composed of a recognition domain that binds to HER2, a marker found on certain kinds of cancer cells, components from the hinge and transmembrane domain of CD8a ( a T cell co-receptor), and a CD3z intracellular domain that is responsible for signaling to the cell. B) The DNA that encodes the CAR was delivered into macrophages using the Ad5f35 virus. Macrophages that are given the CAR DNA can recognize HER2-expressing cancer cells, such are those that occur in aggressive forms of breast cancer. Created with biorender.com.
Chimeric Antigen Receptors (CARs) have opened an entire new vein of research into therapies for cancer and it is exciting to see new developments in clinical trials. Our immune system is potent and adaptable and, with a little help from some clever engineering, we can treat and cure diseases that would otherwise evade detection.
This article summarizes work from Klichinsky, M., Ruella, M., Shestova, O. et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat Biotechnol 38, 947–953 (2020). https://doi.org/10.1038/s41587-020-0462-y
Cover image courtesy of OpenClipart-Vectors from Pixabay