According to the International Agency for Research on Cancer, colorectal cancer is the third most common cause of cancer in the US and the fourth leading cause of cancer – related death worldwide. Research on colorectal cancer has revealed that distinct microorganisms in a patient’s gut microbiota can lead to different colorectal cancer prognosis. Most recently, researchers from the University of Michigan built a system to study distinct gut microbiomes in mice and their potential relationship to colorectal cancer development. Using this system, they found that imbalance of the gut microbiome due to overgrowth of bad microorganisms and too few good microorganisms, led to increased colorectal cancer via inducing dysfunctional immune cells in tumor areas.
The relationship between the gut microbiome and the immune system
A gut microbiome is a group of microorganisms that resides in the human gut. This group is not limited to bacteria only, but also includes virus, fungi, archaea, etc. Humans first acquire their own microbiome at birth from both the mother and the external environment. As we grow up, this group of microorganisms continues to change due to various reasons, such as diet changes, chronic stress, alcohol and antibiotic consumption. Researchers from Harvard Medical School found that changing the microorganisms that make up the gut microbiota can stimulate immune cells. Specifically, the gut microbiota can activate immune cells called CD8+T cells; These immune cells recognize and kill infected or diseased cells. CD8+T cells are potential warriors in the fight against cancer because they can detect and kill cancer cells. However, cancer cells also have some sneaky plans to hijack our immune cells. Tumors can create a harsh environment to overstimulate immune cells and make them exhausted causing them to turn off. By understanding how gut microbes activate the immune system and how cancerous cells can use these activated immune cells for their benefit, we can develop more specific and potent therapies to combat cancer.
Distinct microbiota, different tumor numbers
Taking advantage of a system that has one group of mice with high colorectal tumor numbers (15 tumors) and another group with low colorectal tumor numbers (5 tumors), the researchers decided to examine if there was any difference in gut microbiota and immune cells between the two groups. The two mouse groups had distinct gut microbiota (termed microbiota #1 and #2), and the mice with higher tumor numbers had more immune cells in the colon. There were distinct microbes that made up each microbiota. Microbiota #1 contained microorganisms that can suppress immune cells, while microbiota #2 contained microorganisms that can activate immune cells. Therefore, the distinct microorganisms found in the two microbiota can regulate the immune system differently, revealing one potential reason for the difference in tumor numbers between the mouse groups.
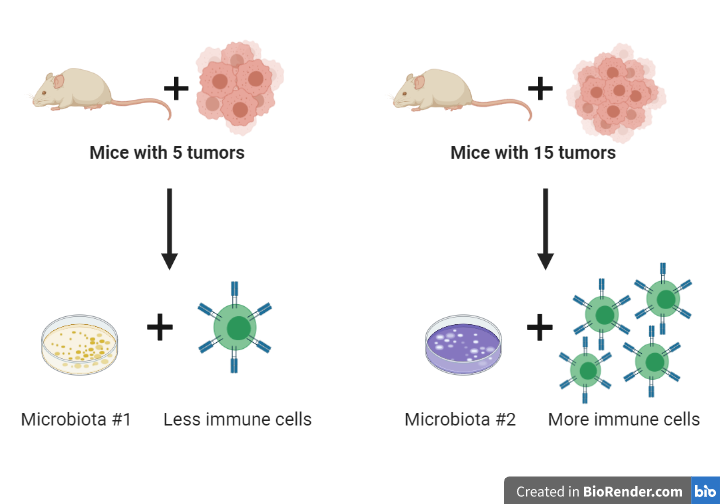
What does the microbiota really do to the immune system?
Since higher tumor numbers were associated with increasing immune cells and distinct microbiota, the researchers examined the immune cells in the entire colon. Their analysis showed increased CD8+T cells in mice with microbiota #2, compared to mice with microbiota #1. To confirm that the distinct microbiota led to different CD8+T cell numbers and different colorectal cancer prognosis, the researchers deleted CD8+T cells in the mice and studied if there was any difference in tumor numbers between mice. Without CD8+T cells, tumor numbers were increased in mice with microbiota #1, while mice with microbiota #2 had decreasing tumor numbers. Therefore, the researchers believe that microbiota #1 induces CD8+T cells’ protective role to cancer, but microbiota #2 regulates CD8+T cells to promote cancer. This finding suggested that tumor numbers were regulated by the distinct microbiome through CD8+ T cells.

Since the main function of CD8+ T cells is detecting and attacking cancer cells, the researchers decided to investigate how microbiota #2 suppresses the CD8+ T cell response. They found that there are a lot of CD8+ T cells in the beginning stage of cancer. However, this number of CD8+ T cells decreased and lost their functions when the tumor formed. This result demonstrated that microbiota #2 increases CD8+ T cells at the beginning of cancer development, but causes reduced CD8+ T cells’ responses within the mature tumor microenvironment and the surrounding normal tissue. The researchers speculate that microbiota #2 may overstimulate CD8+ T cells causing them to enter an exhausted state, at which the CD8+ T cells will lose their functions.
Potential therapeutic treatments
This study has proposed a role of the gut microbiome in altering CD8+ T cells’ activity that can lead to different tumor numbers and cancer prognosis. This finding opens a new perspective in cancer immunotherapy, in which scientists should begin to consider the role of the microbiome in diagnosis and treatments. The authors also underscore the importance of optimizing microbiome transfer methods to determine the microbiome’s specific effects and phenotypes in chronic diseases. Ongoing studies are expected in the future, which is vital for further understanding of the role of the microbiome in cancer treatments.
Journal article: Yu, Amy I et al. “Gut Microbiota Modulate CD8 T Cell Responses to Influence Colitis-Associated Tumorigenesis.” Cell reports vol. 31,1 (2020): 107471. doi:10.1016/j.celrep.2020.03.035
Cover Image: Gut microbes, CC BY-NC
My daughter has tumors in her stomach and we can’t find help or anyone that’ll help her with surgery neither we’ve seen docs in ks and San Antonio tx as well and its been two yes now and shes lost so much weight and can hardly eat at all … the tumors are sticking out of her left side if her stomach and has started forming on the right side as well now you can feel them all and she’s constantly throwing up and in so much pain . We need answers and solutions fast or shez gonna die . I’ve watched her slowly get sicker the last 2yrs now and the last 8months I’ve really watch her get rapidly worse each. Day … shes had 2 biopsies done and mri ultra sound and ea biopsy only made it worse and rapidly spread and get bigger … ea doc. Has told us they cant help her and have exhausted everything they can do to help her and we should see a different specialist and doc. My daughter is 20 yrs.
LikeLike
If you are able to shed light or help her plz call me or email me .. plz asap jlbishop01@gmail.com 3165180971 cell thanks . Thanks. Tumors in my daughters stomach.
LikeLike