As summer slowly approaches, we are going back to the streets after several weeks of quarantine. And with the resumption of activities, air pollution is likely to increase to its former levels. In addition to negative environmental effects, air pollution has been linked to many health problems. For example, a component of polluted air termed particulate matter has a bad reputation for aggravating asthma, although the reason for this has been a mystery until recently.
In a study published in January, researchers found a novel link that could explain how particulate matter worsens asthma symptoms. They report that very small particulate matter (2.5 microns or less in diameter, termed PM2.5) aggravates asthma symptoms by influencing the identity of immune cells.
Inflamed by particulate matter
Particulate matter is the mixture of solid and liquid particles that are suspended in the air we breathe, either of natural (erosion) or anthropogenic (industries, exhaust gases, smoking, etc.) origin. The reason that PM2.5 is so dangerous for human health is that it is very small and light, allowing it to remain in the air for several weeks and easily enter our lungs, followed by our bloodstreams.
Particulate matter is known to worsen many heart and lung-related health conditions, including asthma. Asthma is a chronic disease characterized by breathing difficulties, and while it’s causes are unknown, it is associated with an overzealous immune system that causes inflammation in the airway. This airway inflammation has also been linked to inhaled particulate matter, but the connection between particulate matter and immune activation was unclear. To investigate this relationship in more depth, researchers first tested the effect of PM2.5 in a mouse model of asthma. They injected PM2.5 into the trachea of healthy mice (control group) and asthmatic mice (experimental group). It aggravated asthma symptoms in asthmatic mice, with an increase in the recruitment of inflammatory immune cells to the lungs and increased mucus production.
More precisely, when examining the lungs of the asthmatic mice treated with PM2.5, the researchers found an imbalance in the proportion of two types of immune cells: more Th17 lymphocytes and fewer regulatory T lymphocytes (Tregs). Th17 cells have a pro-inflammatory role that allows them to fight pathogens. Tregs, on the other hand, are anti-inflammatory cells. Their mission is to ensure that the immune system does not get carried away by reacting to harmless foreign entities, such as allergens, or by neighboring “self” cells. Balance between Th17 and Treg cells — between inflammation and its repression — is crucial. Not enough or inefficient Th17 cells would be linked to a less effective body defense against microbes; but on the contrary, under-representation of Treg cells could lead to autoimmune diseases where the immune system becomes overactive and attacks the body. A disruption of this balance, with a higher Th17/Treg ratio, is thought to be involved in severe human asthma, offering one explanation for the abnormal airway inflammation associated with this disease.
Th17 cells and Tregs are of particular interest because they share a common past. Initially, they are naive CD4 lymphocytes, a type of adaptive immune cell. When these naive lymphocytes come into contact with different chemical messengers in their environment, they express, or turn on, sets of genes that predispose them to become either Th17 and Treg cells. Then, secondary chemical messages will seal their fate, differentiating them into one cell type of the other (Figure 1).
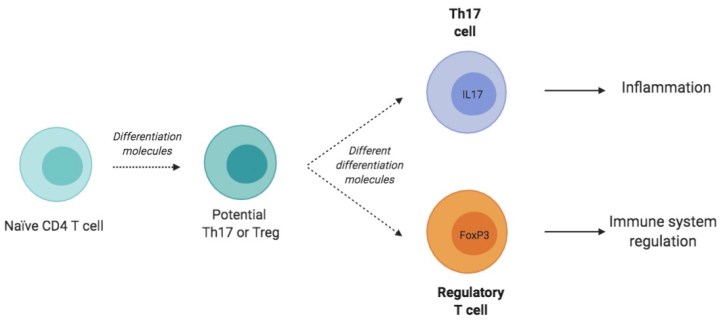
Altering the identity of immune cells
Since the Th17/Treg ratio is unbalanced in mice in the presence of PM2.5, the authors next investigated whether PM2.5 could directly influence the fate of naive CD4 T cells.
Naive CD4 T cells were recovered from mice and grown in media containing molecules that drove them to become either Th17 or Treg cells. Researchers then added increasing concentrations of PM2.5 to the media and analyzed the cells 5 days later. And bingo! The higher the concentration of PM2.5, the more CD4 T cells produce IL17, the main chemical marker of Th17 cells, and the less they produce FoxP3 protein, the main marker of Tregs.
These results indicate that PM2.5 alters the fate of naive CD4 T cells and the Th17/Treg ratio. An imbalance in favor of Th17 inflammatory cells may explain the more severe asthma symptoms. But what happens inside the naive CD4 T cells when they meet PM2.5, and how does this meeting change the fate of the cell?
AhR, a key switch?
To find out, the researchers were interested in a protein present inside many immune cells: AhR (aryl hydrocarbon receptor). This protein is a transcription factor, meaning that it has the power to modify the expression of genes by binding the DNA just in front of the gene sequence. This binding can turn genes on or off. AhR captured the scientists interest because it is known to induce the differentiation of naive CD4 T cells into Th17 or Treg cells, depending on which accessory molecules activate it.
The authors of the study used a new mouse model to find out if AhR is involved in PM2.5-mediated inflammation. They injected PM2.5 into asthmatic mice in which naive CD4 T cells no longer possessed AhR. While PM2.5 normally aggravates asthma symptoms, it did not induce inflammation in the absence of AhR. Furthermore, the lack of AhR led to unaltered levels of IL17 and FoxP3, indicating that the Th17/Treg ratio was no longer disturbed.
These results indicate that PM2.5-induced symptoms and the Th17/Treg imbalance are mediated by AhR. In other words, AhR could act as a switch: being activated in the presence of PM2.5, the fate of the cells shifts towards the Th17 pro-inflammatory identity (Figure 2).
Multiple chain reactions lead to a change in cell identity
Going further in their analysis, the authors found that the PM2.5-induced AhR activation acts at two different levels to influence cell fate. First, it affects the way the naive CD4 T cells produce the necessary energy for their new identity. The PM2.5-induced AhR activation promotes the use of glucose as the cell’s primary energy source; and importantly, glucose metabolism is known to differentiate naive CD4 T cells into Th17 cells.
Secondly, they observed that the PM2.5-induced AhR activation results in deactivation of the gene encoding FoxP3, the protein marker necessary for Treg identity, explaining why fewer Treg cells were present when the mice were treated with PM2.5 (Figure 2).

Balance is the name of the game
In sum, PM2.5 aggravates asthma symptoms in a mouse model, due to its activation of the transcription factor AhR. This specific activation favors the differentiation of naive CD4 T cells into Th17 cells, to the detriment of Tregs. The accumulation of these pro-inflammatory cells could be one of the origins of the asthmatic inflammatory phenomenon.
Interestingly, the researchers also found that PM2.5 modifies the differentiation of human naive CD4 T cells to Th17 cells’ advantage, indicating that their observations could be transposed to asthma in humans.
All processes within a healthy body are finely regulated, and even the slightest disturbance can lead to major disorders. This study shows how a single molecule can disrupt the fate of a cell and, consequently, the health of an individual. Also, this summer, don’t complain too much about the rain that seems to spoil beautiful days. Thanks to rainy days, particulate matter falls back to the ground and will not go directly into your lungs.