How did fish get so lucky? Their oils are revered for their innumerable health benefits while oils derived from the plants that dominate our diet, like corn and soybeans, are distressing to dietitians and immunologists alike. But why the discrepancy? Fish and plant oils are composed of a fatty acid mixture, specifically poly-unsaturated fatty acids (PUFAs), which have minimal bioactivity in mammals like us. They can, however, be broken down to yield molecules called oxylipids, and oxylipids have many duties in the mammalian system. These oxylipids can be potent mediators of inflammation and facilitate many of the positive or negative effects of PUFAs in our diet.
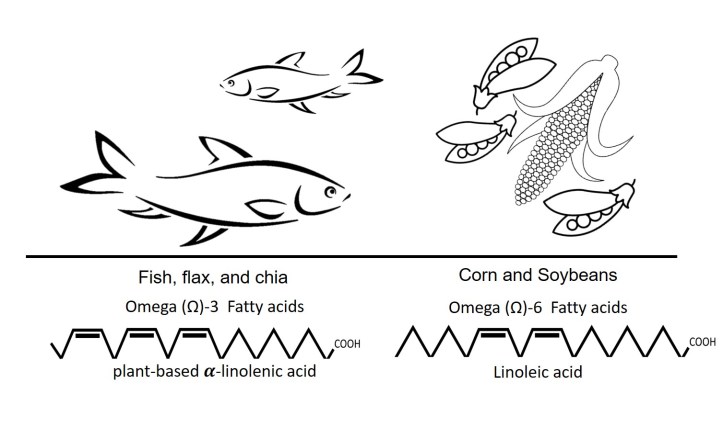
The determining factor between fish-derived PUFA and those that dominate our diet is simply the placement of the first double bond in the chemical structure of the fatty acid. This alteration distinguishes the favorable Omega (Ω)-3 (fish oils EPA and DHA, and plant-based 𝜶-linolenic acid) from the questionable Ω-6 fatty acids (primarily linoleic and arachidonic acids) (Figure 1). While the chemical difference is quite small, the human health impact is quite substantial. This has led the American Heart Association, American College of Cardiology, and European Society for Cardiology all recommend fish oil supplements or weekly fish intake to improve heart health.
There are over 150 Ω-3- and Ω-6-derived oxylipids known to exist, each of which has unique impacts on many health conditions- notably on the cardiovascular system (1). A dizzying field of research has sprung up to study these impacts and attempt to determine if any may serve as biomarkers of disease. For example, taking baby aspirin is a well-known way to reduce the risk of heart attack. Aspirin inhibits the enzymatic production of thromboxanes, oxylipids whose high concentration predisposes people to cardiac events.(2) As a biomarker, the epoxyeicosatrienoic acid (EET) class of oxylipids clues doctors in to how severe the damage of a stroke may be.(3) In these ways, oxylipids act as both biomarkers of disease and therapeutic targets for intervention.
Cardiovascular and ischemic effects cause immune and systemic cascades, some mediated by PUFA, that have serious- sometimes lethal- consequences. In their recent publication, Fang et al. shows why increasing Ω-3 fish oils in your diet can be beneficial heart health by decreasing the long-lasting damage that heart attacks may cause.
Using mice as a model for human heart attacks, Fang et al. identified the oxylipids that were changed during simulated heart attacks. They reasoned these oxylipids were the most likely to mediate the detrimental effects of heart attacks. The authors hypothesized that the protective effects of an increased ratio of Ω-3 to Ω-6 fatty acids in animals would be related to minimizing changes in these specific oxylipids during a heart attack. In other words, a higher Ω-3:Ω-6 ratio would protect the heart from detrimental effects.
After identifying their oxylipid targets, researchers fed mice either a diet supplemented with high amounts of Ω-3 fatty acids or had a gene inserted into their cells that would allow them to convert Ω-3 fatty acids from Ω-6 fatty acids (a trait normally not found in mammals).
The authors looked at several outcomes of simulated heart attacks to determine the overall severity. The outcomes focused primarily on the heart function – how well could it fill with blood and how efficiently it then pumped that blood out of the heart. The most telling outcomes were how well a chamber in the heart could expel the blood it had filled with (ejection fraction) and the distance wall of the heart move during contractions (fractional shortening). Each of these measures are significantly reduced by myocardial infarctions. They also looked directly at the tissue using histology, to determine how widespread the damage (ischemic area) was. Typically, severe damage is correlated to larger ischemic areas.
What they found was encouraging, yet unexpected. Both treatments increased the heart’s ability to contract and expel a relatively greater amount of blood in hearts that had undergone simulated heart attacks compared to untreated groups undergoing the same procedure. Each treatment also reduced the severity of damage caused by a simulated heart attack. These outcomes were promising, but mostly anticipated.
The surprising bit is this: despite both treatments increasing the Ω-3 fatty acid ratio, they did not change the oxylipid target profiles in the same way. The two different techniques to increase the Ω-3 fatty acid ratio, diet vs. gene insertion, each impacted oxylipids identified previously as important to mediating heart attack damage differently. Shifts in oxylipid profiles resulted in positive outcomes from each treatment, but the positive changes were not dependent on specific oxylipids. Rather, broader class shifts were responsible for reducing damage. This outcome means that it may not be enough, or entirely important, to alter any single oxylipid to reduce the severity of a heart attack.
This suggests that we should take a much broader view of the effects oxylipids may have on disease as many are known to be impacted by oxylipids, such as atherosclerosis, localized infections, or pain in general. Instead, due to the significant amount of redundancy between oxylipids, it may be more beneficial to focus on shifting the entire pool of oxylipid production, regardless of the impact on specific oxylipids. Think of it as rebuilding a car. In terms of maintenance and longevity, it would be much more beneficial to slightly upgrade numerous components of a vehicle rather than spend all your money on upgrading the engine alone.
Promisingly, this approach has shown positive results in veterinary science. Lameness in ungulates is typically caused by inflammation within their hoof and NSAIDs are typically, although generally ineffectively, used to reduce this inflammation. Dr. Hammock’s team focused on using a compound that inhibits the enzyme Soluble Epoxide Hydrolase, which is responsible for producing a family potently pro-inflammatory oxylipids known as DiHOMEs. By blocking the production of DiHOME oxylipids, inflammation was reduced in the horses, decreasing their pain and lameness (4,5). While not a cardiovascular disease, this research underscores the breadth of impacts oxylipds have on health.
In humans, we can potentially manipulate the ratio of oxylipid families by increasing the abundance of Ω-3 fatty acids in our diet- ideally by consuming more Ω-3 rich foods. Ω-3 rich foods include seafood, flaxseed, chia seed, eggs or milk fortified with Ω-3s, and cruciferous vegetables. While diet and exercise are always a positive course of action to maintaining heart health, incorporating Ω-3 fatty acids into our diets may be a viable way to further reduce the incidence and severity of heart disease.
Article: Prostaglandins Other Lipid Mediat. 2018 Nov;139:1-9. doi: 10.1016/j.prostaglandins.2018.09.002. Epub 2018 Sep 14.
CITATIONS:
- Advances in Our Understanding of Oxylipins Derived from Dietary PUFAs. Gabbs, M et al. 2015.
- Low-Dose Aspirin for the Prevention of Atherothrombosis. Patrono, C, et al. 2005.
- Epoxyeicosatrienoic Acids are Mediated by EPHX2 Variants and may be a Predictor of Early Neurological Deterioration in Acute Minor Ischemic Stroke. Yi, et al. 207.
- Soluble epoxide hydrolase activity and pharmacologic inhibition in horses with chronic severe laminitis. Guedes, et al. 2017
- Use of a soluble epoxide hydrolase inhibitor as an adjunctive analgesic in a horse with laminitis. Guedes, et al. 2013