When thinking about the 80’s, the first things that come to mind are probably brightly coloured spandex and questionable perms, not an epidemic. Although HIV was eventually detected in samples dated over a decade prior, it was only during the 1980’s that the true potential of this virus came to light.
After an unexplained immunodeficiency emerged across the gay community in the USA (later diagnosed as acquired immune deficiency syndrome, or AIDS), the causative agent, human immunodeficiency virus 1 (HIV-1), was isolated in France in 1983. As seen with COVID-19, understanding the effects, transmission, and progression of a brand new virus can be limited and hypothetical, as true understanding can only be achieved by data which takes time to collect. The limited knowledge available nurtured misunderstanding and consequently, the retrovirus HIV was only thought to affect gay men, intravenous drug users, and recipients of contaminated blood transfusions. With transmission routes unknown, a sense of fear was generated amongst the public and, even in the present day, HIV is still stigmatized. Thankfully, scientific research has had a propelling impact and perceptions of the virus are changing. Although there is no cure, successful treatments and preventative measures are available, and further research means that new therapeutics may be on the horizon.
How is HIV transmitted?
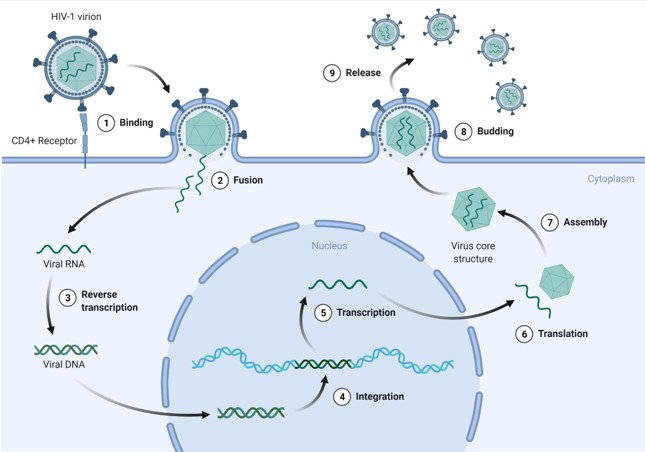
The routes of transmission are varied and include intercourse, use of contaminated needles, and transfer from mother to child. 80% of HIV-1 enters via the vagina or rectum through the mucosal membranes, which is why a large focus of HIV awareness campaigns are directed at sexual prevention. To access the bloodstream, the virus needs to pass through epithelial cells that line these membranes. There are a number of these epithelial cell layers in the vaginal and penile areas, but damage to these layers (e.g. during sex) can increase the viruses chances of transmission. However, in the rectum, this epithelial layer is only one cell thick; this means that there’s less of an external barrier to viral entry and explains why HIV infection tends to be more prominent among men and transgender women who sleep with men. Once in the bloodstream, the virus is free to infect CD4+ T cells, a type of white blood cell that plays a large part in the immune response. With its target in sight, the viral particles are able to bind to the host cell and fuse its encapsulating envelope with the membrane of the CD4+ T cell. This action facilitates the release of the virus’s genetic material into the cell, meaning that it can now take full advantage of the host cell’s machinery to make many more copies of itself (Figure 1).
How does HIV become AIDS?
As HIV infects and destroys CD4+ T cells, inevitably, over time, the overall number of these immune cells will decrease. To be diagnosed with AIDS, an individual’s CD4+ count will have fallen to below 200 cells per cubic millimeter of blood (200 cells/mm3). To put this into perspective, someone without HIV can have a CD4+ number of anything between 500 and 1500 cells/mm3. Left undetected or untreated, HIV will on average take 8 to 10 years to reach this cell count and ultimately, an AIDS diagnosis.
There are a number of different types of CD4+ T cells with a variety of roles within the body. Some are responsible for killing infected cells, others recognize pathogens and stimulate antibody production, and still others have regulatory properties for maintaining a healthy immune system. Involvement from all these subtypes contributes to a well-functioning immune system so, as you can imagine, lower numbers mean an increase in susceptibility to infection. With a weakened immune system, any opportunistic pathogen that may make a healthy person slightly unwell can be devastating to someone with AIDS and can often lead to illnesses like Tuberculosis, Kaposi’s sarcoma, invasive cervical cancer, and herpes. Fortunately, medicine has come a long way since the 80’s and an HIV diagnosis isn’t the death sentence it used to be. Although there’s no cure, it’s incredibly treatable, and preventative measures are available.
AZT
One of the first go-to treatments in the 80’s for people with HIV/AIDS was a drug called azidothymidine (AZT). Featured in films such as the 2013 Dallas Buyers Club, which followed Matthew McConaughey as Ronald Woodruff and his journey after an AIDS diagnosis, AZT was initially developed as an anticancer drug. Although found to be effective against viruses in mice, it failed when it came to human cancer trial participants and was shelved. However, when the HIV epidemic arose, there was a need to find a treatment, and fast. Instead of years, AZT became FDA approved in a mere 20 months and is still one of the quickest drugs to gain treatment approval to date.
To bring it to market, a pharmaceutical company and scientists teamed up to assess the effect that AZT has on HIV-infected T cells. The researcher leading this, Dr Samuel Broder, reported that, during the first stages of this testing, AZT appeared to protect CD4+ T cells from HIV-1 by preventing viral replication, suggesting that it could be an effective treatment for HIV infection. This success was also observed again during a human trial study where 19 of the 282 AIDS or related patients died compared to 1 receiving AZT, with researchers deeming that the trial wouldn’t be ethical to continue. It’s still one of the antiretroviral treatments used today, usually in combination with another (HIV mutates often, so combining antivirals can decrease the risk of a drug resistant strain developing).
CRISPR
Antiretroviral treatments can prevent viruses from replicating, but HIV can lay latent and resurface if treatment ceases. New developments to tackle this include the ability to edit genomes through clustered regularly interspaced short palindromic repeats (CRISPR). There has been some buzz around CRISPR’s potential for a while, but a company called Excision BioTherapeutics has recently received FDA clearance to begin the first-in-human Phase 1/2 clinical trial.
Known as EBT-101, the CRISPR-Cas9 system allows specific DNA segments to be snipped from invading pathogens and used as memory tools. If invasion from the same pathogen occurs the DNA can be cut, therefore destroying the genetic material.
When HIV infects human cells, it integrates its DNA into ours, making it very difficult to eradicate by traditional methods (Figure 1). This new CRISPR-Cas9 technology has been engineered to remove the viral DNA from the human genome and insert new DNA in place of the HIV sequence. Originally evolved as a bacterial survival mechanism, this genome editing therapy is specifically programmed, causing no harm to important parts of the human genome. In addition, CRISPR elicits a very low immune response, making it viable for gene therapy, and hasn’t been known to cause any other pathologies so far.
PrEP
Ultimately, the best approach to HIV management is prevention. Awareness and barrier contraception are generally good for low risk individuals, but for those whose risk is higher, other options are necessary. Truvada is one of the main pre-exposure prophylaxis (PrEP) drugs and can be used effectively against all types of transmission. A type of combination therapy, the drugs emtricitabine and tenofovir are taken in a daily pill form and have been found to decrease the risk of sexually transmitted HIV infection by 99% when taken as prescribed.
Potential Vaccine?
There may also be a vaccine in the not so distant future, as Moderna is set to begin human trials for their mRNA vaccine. One of the long term common challenges of vaccines is that as viruses mutate, the immune system is no longer able to recognise and destroy virus particles. However, Moderna has created one that produces antibodies targeted at parts of the virus’s envelope that largely remain unchanged after mutation. This can also be modified to account for new variants so if it proves to be effective in trials, this could be a huge breakthrough for those in at-risk communities.
Although there are challenges ahead, a better understanding and advances in medicine mean that the future looks promising and a diagnosis may one day result in a cure.
Cover image by mohamed Hassan from Pixabay