This time of year we constantly hear about fever, whether it be due to flu season or the ongoing COVID-19 pandemic. Come to think of it, fever is a very common symptom involved not only in infections, but also chronic inflammation and autoimmune disease. Why is that? While fever during infections is characterized as beneficial, the role of fever in autoimmune disease is not well understood.
A recent study reveals a new found role for fever in autoimmune disease. Wang and colleagues present evidence that fever favors a specific population of the critical immune cell called T cells and that this population contributes to the severity of autoimmune disease.
Feeling hot, hot, hot
Fever is a temporary increase in body temperature above the average of 98.6°F (37°C) and has been conserved in vertebrates for millions of years. Normally, fever can provide us with a survival advantage by creating an unfavorably hot environment for invading pathogens and by boosting our immune system, which can function more optimally at a slightly elevated temperature. However, this amped up immune response can cause harm when it goes unregulated, which is the case in autoimmune disease.
Autoimmune disease occurs when the body’s immune system is too active and starts attacking its own healthy tissue. Fever is a major clinical symptom of many autoimmune diseases. In fact, about 20% of patients that have fever from unknown origins are later diagnosed with an autoimmune disease. Treatment with drugs that have anti-fever properties, like aspirin, reduces immune responses causing inflammation and alleviates symptoms in patients. This indicates that fever, which is meant to help us, is somehow contributing to the increased immune responses and severity of disease in patients. Despite its commonality, the role of fever in autoimmune disease has not traditionally been well understood.
To try and understand this, the researchers in this study wondered if fever had any effect on the important immune cell called T cells and if this effect contributed to autoimmune disease severity.
The many, mighty helper T cells
Our immune system has two major classes of T cells: cytotoxic T cells and helper T cells. This paper focuses on helper T cells, also called CD4+ T cells. Consistent with their name, helper T cells function to regulate other cells of the immune system through production of small molecules called cytokines. Cytokines are chemical messengers, similar to hormones, that carry messages from one cell to another, instructing the cell’s next actions. There are multiple classes of CD4+ T cells that are classified based on their production of specific cytokines. Some of these subsets include T helper 1, T helper 2, T helper 17, and T regulatory cells. These different subsets have varying functions, whether it be fighting infection, wound healing, or immune system regulation.
Regardless of their particular job in the body, it is vitally important that the activation of these various T cell subsets is tightly controlled. We want them to be active, for example, when we’ve been infected by a virus or bacteria so that they can eradicate the threat. But, we do not want them to be active in the absence of a threat, as overactive immune cells can attack healthy tissue, leading to chronic inflammation and autoimmune disease. For example, previous studies have found that excessive T helper 17 (Th17) cell responses contributed to chronic inflammation in patients with inflammatory bowel disease.
In this new work out of Tsinghua University in Beijing, scientists studied the effect of fever on the various classes of CD4+ T cells and if this contributed to autoimmune disease development.
Th17 Cells like it hot
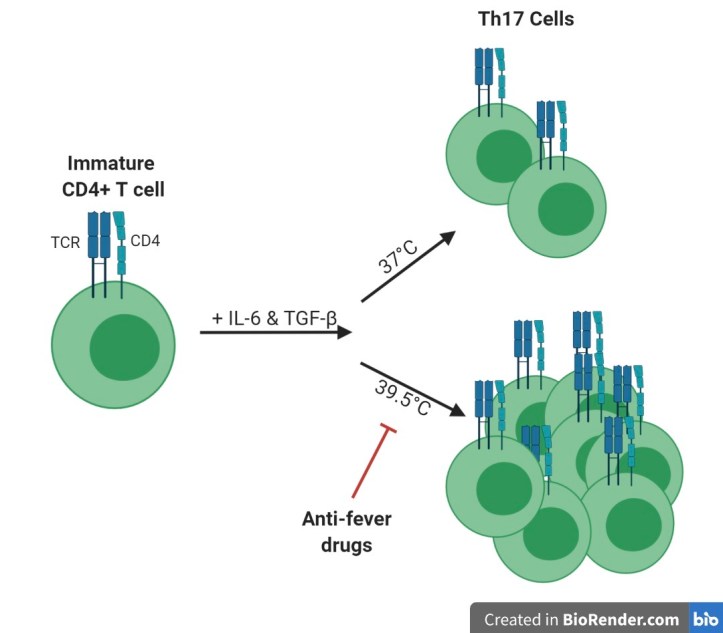
The researchers first wanted to see how fever affected the development of the various classes of CD4+ T cells. They grew immature CD4+ T cells in a dish containing specific cytokines to drive them to become each specific class of CD4+ T cell. For example, an immature CD4+ T cell needs to interact with cytokines IL-6 and TGF-β to become Th17. They grew the cells at either normal temperature (37°C) or fever-like temperature (39.5°C). They found that only Th17 cell populations were enhanced under fever-like temperature, while the other subsets were unaffected by the temperature increase (Figure 1).
Next, the researchers tested the effect of fever on Th17 cells in a mouse model of autoimmune disease that specifically mimics multiple sclerosis. When this model was induced, the mice developed fever and severe symptoms, particularly paralysis of their limbs. In these mice, Th17 cell populations were increased and production of Th17-specific cytokines was increased as well. As mentioned above, cytokines act as messenger molecules. One common message conveyed is a summons to other immune cells. So, some of the Th17-specific cytokines can call additional immune cells into tissues. Consistent with this idea, these mice had increased recruitment of other immune cells into their tissues, creating a dangerous environment filled with an arsenal of immune cells capable of attacking healthy tissue. Treatment with anti-fever drugs, like aspirin or ibuprofen, reduced these enhanced Th17 responses.
These studies show that fever can favor the activation of Th17 cells and could be one reason why excessive Th17 cell responses have been seen in patients with inflammatory bowel disease and multiple sclerosis for example.
What’s the SMAD-er with you?
Next, the researchers wanted to know what specifically happens inside of an immature CD4+ T cell under fever conditions to drive these enhanced Th17 cell responses.
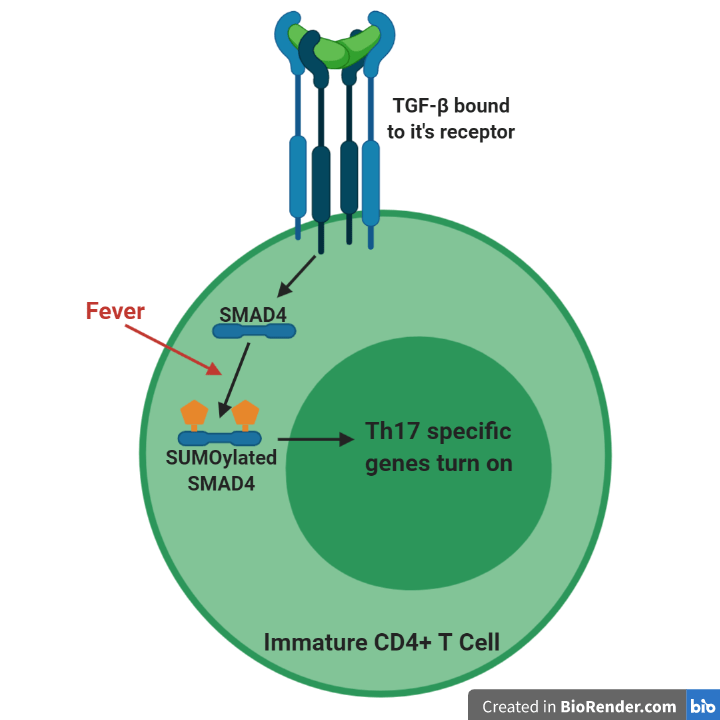
For an immature CD4+ T cell to become a Th17 cell, it needs to interact with the specific cytokines, IL-6 and TGF-β, in the nearby environment. Once these cytokines bind at the surface of an immature CD4+ T cell, molecules inside the cell called transcription factors get activated. These transcription factors are responsible for turning on genes which make the immature CD4+ T cell a Th17 cell. One type of transcription factor important for Th17 cell activation is SMAD4, which turns on when the cell binds TGF-β. Transcription factors have many different roles inside a cell, and oftentimes, their job description can change by the addition of small molecular tags. Just like a cookie can change by the addition of chocolate chips or raisins, transcription factors change when different tags are attached to them. One type of tag that can be added is called SUMOylation.
The scientists decided to test if SMAD4, and specifically SUMOylation of SMAD4, was required for the enhanced Th17 cell responses observed during fever-like conditions.
Through a series of elegant studies, the researchers found that under fever-like conditions, SMAD4 SUMOylation was enhanced which was necessary for the enhanced Th17 cell populations and severity of disease.
The most notable experiments were the ones conducted in the mouse model of multiple sclerosis. First, they induced disease in either normal mice or mice that lacked SMAD4 specifically in CD4+ T cells. Mice lacking SMAD4 in their CD4+ T cells had delayed disease onset, reduced disease symptoms such as paralysis, and a reduced number of Th17 cells in the central nervous system. This showed that SMAD4 was necessary for the enhanced activation of Th17 cells and disease progression. Next, they performed similar experiments in which they used either normal mice or mice that had CD4+ T cells with SMAD4 that could not get SUMOylated. Mice with the SMAD4 that could not get SUMOylated also had less severe disease and reduced Th17 cells in the central nervous system. This showed that, not only SMAD4, but specifically SUMOylated SMAD4 is required for the enhanced activation of Th17 cells under fever conditions.
Overall, these studies discovered that fever increases the SUMOylation and activation of the SMAD4 transcription factor which causes increased Th17 cell populations to appear (Figure 2). The resulting increased Th17 cell populations contribute to more severe autoimmune disease in a mouse model of multiple sclerosis.
It’s all about the balance
So, what’s the answer? Is fever good or bad for us? Well, it’s all about the balance. Fever certainly is helping us when we have some kind of infection. Here, we learned that fever can specifically enhance Th17 cell responses. The key is tight regulation. The immune system functions best when there is tight regulation between activation and suppression of immune responses. When something goes wrong with this balance, your immune system can start to attack your body resulting in an autoimmune disease. The chronic fever that comes along with these diseases can cause more robust immune responses contributing to disease severity. As seen in this study, fever induced an enhanced Th17 cell response which contributed to the severity of autoimmune disease. Autoimmune disease patients oftentimes take immunosuppressants. This is a harsh treatment that can cause devastating long-term side effects and leave patients highly susceptible to harmful infections. The search for alternative treatments continues. While fever reducing drugs are already used for autoimmune disease patients, perhaps their clinical use could be re-evaluated and adjusted based on studies such as these.
Cover image: Fever, child, common cold; CC BY-NC-SA 3.0
Journal article: X. Wang, L. Ni, S. Wan, X. Zhao, X. Ding, A. Dejean, C. Dong. Febrile temperature critically controls the differentiation and pathogenicity of T helper 17 cells. Immunity, 52 (2020), pp. 328-341. https://doi.org/10.1016/j.immuni.2020.01.006