Take yourself back to a visit to an aquarium. Likely, you were in awe of the diversity of fish swimming in the tanks before you – a dazzling array of colors, shapes, and sizes. Certain species of fish date back hundreds of millions of years, including jawless fish like the sea lamprey. Sabyasachi Das, Yoichi Sutoh, and their colleagues at Emory University and the University of Pennsylvania were interested in the similarities between lampreys and other animals with respect to their immune systems. In an article published in The Journal of Immunology this fall, this research group discovered two lamprey genes involved in immune cell signaling which were very closely related to those present in jawed fish and other vertebrates. Their research indicates that specific immune cell signaling pathways evolved prior to the point in time when jawless and jawed fish diverged on the evolutionary tree of life. This means that ancient fish, like lampreys, often considered to have a more rudimentary immune system, may not be as different from humans in this realm as previously thought.
Our immune systems are extremely complex, employing a wide variety of cells and proteins to effectively fend off disease causing organisms. Many of these cells have very specific roles to play and respond to specific signals from other cells and their environment. One can think of all the different cell types as actors in a theatrical play. Certain cells and proteins make their appearance in Act I, setting the stage and providing initial information about the plot. This is analogous to our innate immune system, which is a broader response to invading organisms and is less specific. Others wait in the wings until Act II. At this point, the plot is clear – your immune system has learned what invading agent is present in your body and wants to contain that agent, wrapping up the story. The cells involved in this later response to infection are a part of your adaptive immune response. These cells are primed with knowledge about what invading organism is present in your body, and use their collaborative power to eliminate that threat.
One cell type involved in this adaptive response is the B cell. B cells have to receive complex signals in order to become activated, differentiate into mature forms, and become equipped to appropriately respond to foreign invaders. It is the signals that are involved in creating functional B cells that drew the attention of Das, Sutoh and their colleagues within the sea lamprey. Specifically, they noticed that sea lampreys have genes that were strikingly similar in sequence to B cell signaling molecules that have been well characterized in higher order vertebrates, such as humans, mice, and dogs. These include B cell activating factor (BAFF) and the B cell maturation antigen (BCMA) receptor, both of which help maintain populations of B cells capable of producing antibodies. Antibodies are protein molecules that can specifically bind to invading organisms, targeting them for destruction or signaling to other cells that there is an immune threat.
Das, Sutoh and colleagues used DNA that they obtained from cells within lamprey blood to investigate similarities in genetic sequences. After undergoing extensive analytic comparisons between species ranging from sharks to cows, they were able to connect two genes in the lampreys to ones that encode for the BMCA receptor in jawed fish and other vertebrates. A deeper dive into the structure of these BMCA-like genes revealed that they contain sites capable of binding to a BAFF-like molecule previously described in lampreys. Further, when they constructed three-dimensional models of the lamprey BMCA-like receptors and compared them to the structure of the human BMCA receptor, they noticed that they were similar in structure and electrostatic charge. This demonstrated that an ancient fish has similar B cell signaling to animals that evolved much later in Earth’s timescale. Remember, lampreys have been around for 360 million years! For a cell signaling process to be highly conserved throughout that length of time speaks volumes to the importance of B cell signaling to the functionality of the immune system, and to the preservation of healthy living things.
The researchers weren’t satisfied with knowing that the lamprey genes were similar in sequence and structure, they wanted to know if the proteins produced from those genes were functionally similar to those in more advanced species, like humans. In the laboratory, the researchers were able to grow cells which contained the lamprey BMCA-like receptors, and showed that those cells could indeed bind to the lamprey BAFF-like protein. This similar binding interaction is observed in human B cells and is important to ensuring that B cells act appropriately in their role in the immune system play. Additionally, within the lamprey, the highest amount of the BMCA-like receptor was found in circulating blood cells, and more specifically, on the surface of the lamprey B-cell equivalent. The two BMCA-like genes were also implicated in regulating different steps of the B cell maturation process; one was observed mostly within cells that had never been exposed to a stimulus, while the other was observed within stimulated cells. This is similar to human B cells, which express a different repertoire of receptors on their surface when they have never seen an infectious agent versus when they have been exposed to one. It is these changes in B cell surface receptors that allow the B cell to become a better actor in the adaptive immune system, becoming more specific to the invading organism and producing large amounts of antibodies. Certain populations of B cells will even stick around after the infection has been cleared, and serve to help remember your previous exposures.
These findings suggest that interactions between BAFF-like and BMCA-like molecules evolved prior to the divergence of jawed and jawless vertebrate lineages. B-cell signaling was likely critical for ancestral organisms, and thus persisted as higher order animals evolved. This is important, because lampreys and other ancient organisms are often thought of as being more simplistic, or primitive, when compared to other animals. Now, we know that this is not the case, and that there may be more similarities between ancient fish immune systems and our own than has been previously considered. Uncovering similarities between lampreys and other vertebrates in immune cell signaling, like those revealed by this research group, opens the door for lampreys to be used as an animal model in immunology studies. These models allow researchers to answer key questions about immune system function and dysfunction. While additional research is needed to further characterize these molecules and understand how they interact during an infection within lampreys, this study adds critical insight into our knowledge of the evolution of an extremely complex cellular system and emphasizes the strong links between diverse species within the animal kingdom. Sometimes, we have to look to our past to better understand our present.
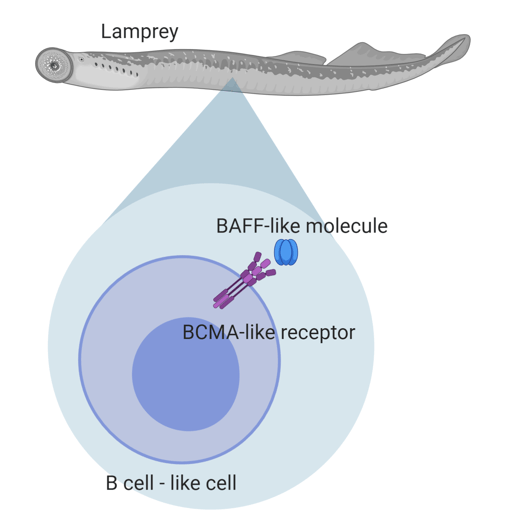
Das, Sutoh, and colleagues discovered gene sequences in the sea lamprey which were highly similar to those encoding BMCA receptors in jawed fish and higher order vertebrates. Through experimental studies, they demonstrated that the lamprey BMCA-like receptors could bind to lamprey BAFF-like molecules, predominantly on lamprey B cell equivalents. This suggests that interactions between BAFF-like molecules and BMCA receptors evolved prior to the divergence of jawed and jawless fish, and have persisted throughout evolutionary history. Image produced by Dilara Kiran using Biorender.
Dilara Kiran is in her sixth year of the Combined Degree DVM/PhD program at Colorado State University. She aspires to use her knowledge of both clinical practice and research to contribute to evidence-based scientific policy and is passionate about science communication. Follow her on Twitter @dvmphd2be.