In 1909, a German scientist named Paul Ehrlich made a simple yet shocking proposal: that cancer arises in our bodies quite frequently, but that it is usually eliminated by cells of the immune system before tumors form. Although radical at the time, Ehrlich’s theory gained experimental support in the mid-to-late 20th century, and it is now widely accepted that immune cells can indeed recognize and eliminate cancer cells. Why, then, is cancer still to blame for an estimated 8.2 million deaths worldwide annually? The answer lies in cancer’s ability to adapt and evolve to evade cells of the immune system. Interestingly, one way that cancer cells achieve this is by commanding “DON’T EAT ME!”.
Recently, a collaborative group of scientists from Stanford reported a novel function for MHC class I as a “don’t eat me” signal. In this previously undiscovered role, MHC class I proteins may influence the ability of cancer cells to form tumors despite the presence of a functional immune system.
“Don’t eat me” signals
The primary job of the immune system is to recognize and kill foreign pathogens, like bacteria and viruses, that pose a threat to our health. Because many immune cells possess potent killing abilities, it is vital that they be able to distinguish foreign, dangerous cells from the “self” cells of our own body. One way that “self” cells communicate their identity is through “don’t eat me” signals, which are proteins expressed on the cell surface that mark the cell as belonging to one’s own body, as opposed to being a foreign invader. The prototypical “don’t eat me” signal is CD47. When CD47 is recognized by its receptor (SIRPα) on the surface of macrophages, these cells are no longer able to effectively engulf, or phagocytose, other cells, and thus the self-cell is not killed.
While “don’t eat me” signals have naturally evolved to prevent immune destruction of “self” cells and thus to prevent autoimmunity, cancer cells have co-opted and exploited this strategy to evade immune recognition and attack. Accordingly, CD47 is expressed on a high proportion of cancers, and its expression is associated with reduced patient survival. Because of this high expression, therapeutic drugs aimed at inhibiting CD47 are currently being tested in clinical trials to assess their anti-tumor efficacy in patients with various types of cancer.
MHC class I: a novel “don’t eat me” signal
Despite the potential of this CD47-targeted anti-cancer therapy, a recent study by Barkal et al. reports that that not all cancer cells are sensitive to drugs that inhibit CD47. In theory, treating cancer cells with a drug that binds to CD47 would prevent the SIRPα protein on macrophages from binding to CD47 (Figure 1). Without this binding, phagocytic processes in the macrophages should not be inhibited, and thus macrophage-mediated cancer cell engulfment should increase. And indeed, Barkel et al. report that this is the case for some cancer cells…but not for all.
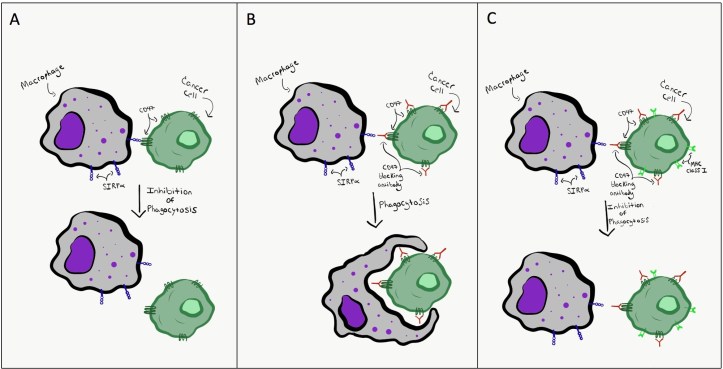
The fact that some cancer cells fail to show more robust phagocytosis by macrophages has an important implication. Specifically, this finding indicates that these cancer cells likely have some other, previously undiscovered, “don’t eat me” signal that is preventing their engulfment, even though CD47 is being blocked. This is precisely what Barkel et al. uncovered: they report that, in addition to their classical role in antigen presentation, MHC class I molecules also function as a novel “don’t eat me” signal on cancer cells.
The paradox of an MHC class I ‘don’t eat me” signal and its therapeutic implications
The role of MHC class I, and the reason that it is so widely expressed, is to display antigens, or immunogenic protein fragments, on the surface of cells. T cells that recognize antigens in MHC class I complexes are capable of killing those cells. MHC/antigen complexes play a vitally important role in alerting the adaptive immune system to the presence of tumor cells, and thus the expression of MHC class I molecules are normally thought to assist the adaptive immune system in killing tumor cells.
And so we are presented with a paradox: MHC class I both facilitates the killing of cancer (by adaptive immune cells) AND prevents the killing of cancer (by macrophages).
These two discordant roles for the MHC class I complex dictate that, evolutionarily speaking, a cancer cell has a choice. Option 1 is to express MHC class I proteins in order to exploit their “don’t eat me” functions. Option 2 is to reduce MHC class I expression, thereby decreasing the number of antigens displayed on the cancer cell surface and thus becoming less visible to the adaptive immune system. In fact, in-depth examination of cancer has revealed that cells tend to lose MHC class I expression over time, meaning that natural selection favors the survival of cancer cells with low MHC class I expression over cells with high MHC class I expression. It follows that, of the two disparate functions of MHC class I, the role of MHC class I in presenting antigens to the immune system is more important for cancer cell survival than is the ability of MHC class I to serve as a “don’t eat me signal”.
This knowledge of cancer’s preference to get rid of MHC class I makes the discovery of Barkal et al. extremely exciting. Specifically, if cancer cells downregulate MHC class I expression to evade the adaptive immune system, they may be inadvertently sensitizing themselves to death-by-macrophage. In other words, if cancer cells have a natural propensity to decrease MHC class I expression, then they open themselves up to macrophage-mediated phagocytosis, a weakness that can be exploited by doctors. For example, a patient whose tumor has lost MHC class I expression might be a great candidate for anti-CD47 therapy, which, as mentioned above, sensitizes cells to killing by macrophages. Thus, by downregulating MHC class I, cancer cells may provide a therapeutic window for exploiting their sensitivity to phagocytosis.
For the original journal article, please see: Barkal, A. A. et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nature Immunology 19, 76–84 (2018).