Zika virus (ZIKV) has emerged as a significant human health threat in the last 5 years. Zika virus is associated with (and may cause) microencephaly and neurological deficits in infants, with unknown long-term consequences for adults. In late 2015 there was a spike in ZIKV infections in South America. Initially, the surge was blamed on the influx of international travelers into and out of Brazil for the World Cup, but viral genealogists determined the virus actually arrived in Brazil in 2013.
The Aedes species of mosquitoes carry and transmit Zika virus. Proteins in mosquito spit have been shown to aid the transmission of other viruses, such as dengue fever—a virus closely related to ZIKV. .
The consequences for human reproduction, wide range of the mosquito vectors that spread ZIKV, and the unknown long-term effects combine to make a ZIKV an immediate and global health concern.
Although a causative link between ZIKV and fetal abnormality has been suspected, brand new research has established the first direct linkages between the infection of pregnant mice and pathological outcomes in mouse pups. A more complete understanding of ZIKV infection opens up avenues for vaccine and treatment therapies.
Lin Jin et al. in Nature Immunology hypothesized that there was a factor present in mosquito saliva which increased ZIKV virulence, and report that a protein in mosquito saliva (LTRIN) aids Zika virus transmission to human cells. The authors show that LTRIN aids infection of human and mouse cells by interfering with two important immune signaling pathways: Lymphotoxin beta receptor (LTβR) and NF-kappa-B.

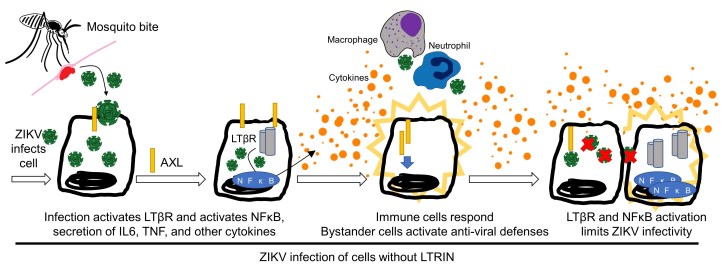
Q: What happens when ZIKV infects target cells?
ZIKV infection of cells activates LTβR signaling, which increases NFκB activation and stimulates the production and release of pro-inflammatory cytokines (IL-6 and TNF) – all of which contribute to controlling and eliminating ZIKV and other viral invaders. Interference with LTβR makes bacterial, viral, and parasitic infections more aggressive or “virulent.”
Viruses normally go through a replication cycle (Figure 1) which, without alteration, would alert the innate and adaptive immune systems to the invasion. Viruses have evolved a number of strategies to fool, silence or otherwise evade our immune systems.
FIGURE 1
Virulence factors are traits and tricks carried by viruses to make them better at infecting hosts. LTβR signaling is a critical step in skin cells alerting the local immune system of the presence of an invader- cut those messages off, and the virus gains an advantage.
Q: Why does the mechanism of Zika virus infectivity matter?
Understanding how a virus alters communication between infected cells, early immune responses, and the immune system at-large may allow for development of treatments for people with Zika, or ways to prevent ZIKV infection.
Q: What do we know already?
Preliminary data suggests that the lymphotoxin beta signaling pathway is altered or blocked to increase ZIKV infectivity. Normally, LTβR is activated by one of two ligands (LTαβ2 or LIGHT), binds into trimers or dimers, and then activates immune molecules such as NFκB. Once activated, the NFκB family of proteins turn on specific genes that alert the immune system to the presence of a viral infection, thereby galvanizing immune-mediated anti-viral responses.
Through a series of logical and well-controlled experiments, Jin et al. demonstrated that mosquito saliva contains an immune-inhibiting protein (LTRIN) that binds preferentially to LTβR, interferes with NFκB signaling, increases ZIKV viral load and enhances viral dissemination within infected hosts.
Q: Does LTRIN have an effect on the behavior of LTβR ?
To test whether LTRIN protein -and not the other proteins in mosquito saliva- could enhance ZIKV infection, the researchers treated murine cell cultures with pure LTRIN protein and ZIKV. LTRIN protein alone prevented LTβR dimerization and the downstream activation of NFκB molecules. Blocking the activation of NFκB prevents normal anti-viral responses, giving ZIKV an advantage over the host
To test if LTRIN had a similar effect in human cells, two cell lines were infected with ZIKV with and without LTRIN. Human-derived cell lines (THP-1 and HUVEC) treated with LTRIN grew more virus per cell (viral load) than cultures with either ZIKV alone or ZIKV + the normal LTβR ligand (LTαβ2). In other words, the presence of LTRIN alone was adequate to boost virulence and viral reproduction in murine and human cells.
Sidebar: How does ZIKV get into a human cells?
The mechanism ZIKV uses to enter human cells has been established. AXL is an indispensable protein in ZIKV infection of human cells. It is a “Tyro3 Axl Mer” (TAM) family tyrosine kinase, which are generally involved in the clearance of apoptotic (dying) cells. AXL also plays an important role in regulating innate immune reactions, including the anti-viral interferon programs. AXL, normally expressed on the cell surface, is up-regulated after initial ZIKV infection. (Figure 2)
FIGURE 2
Q: So how is LTRIN helping ZIKV infect cells?
In both human and mouse cells, the presence of LTRIN increased both the amount and membrane localization of AXL in targeted cells- even in the absence of ZIKV. The increase in this helper protein at the surface likely increases the number of viruses which can enter individual cells, and may explain the increase in ZIKV found in cells co-exposed to ZIKV and LTRIN.
The authors did not (yet) determine the mechanism of AXL increase as a result of LTRIN.
That isn’t the whole story, however. The authors drilled down further, to determine which steps in NFκB activation were changed by LTRIN.
Q: Which part(s) of the NFκB pathway are are hit by LTRIN?
To do this, they used two chemical inhibitors which prevent specific steps in the NFκB pathway. One of the inhibitors (PDTC) blocks the “canonical” or regular NFκB path, and the other (ACHP), blocks the “non-canonical” or alternate NFκB path. They compared these NFκB inhibitors to ZIKV+LTRIN.
All three treatments prevented the creation of pro-inflammatory cytokines and increased virus numbers in target cells. Blocking NFκB activity prevents the cell (and thus the host) from mounting and coordinating an early immune response against ZIKV. Delaying innate immune activation also delays adaptive immune activation, and allows ZIKV a window of uninhibited replication and infection. (Figure 3)
FIGURE 3
Q: Do observations made in a cell culture dish translate to ZIKV infection in an animal model?
The authors used a widely-accepted mouse model of ZIKV pathogenesis (Ifnar-/-). Mice that were given LTRIN at the time of ZIKV infection had higher virus presence (by qPCR) in the brain, spinal cord, liver, kidney, and serum than mice infected with ZIKV alone. Axl (the membrane-bound helper protein) gene expression was also significantly higher in the livers of mice treated with ZIKV and LTRIN. This specifically suggests that ZIKV disseminated to and was in the process of infecting the liver. This is further than the virus and its effects are detected when LTRIN is absent.
These results together suggest a greater rate of cell invasion, viral reproduction, and systemic penetration as a result of the presence of LTRIN.
The overall effects of blocking LTβR activation, and the subsequent blockade of NFκB has profound effects on how far, how fast, and how aggresively ZIKV can infect a host.
Q: Can we block the effect of LTRIN in ZIKV infection directly?
The authors tested whether blocking LTRIN (via an anti-LTRIN antibody treatment) during mosquito feeding altered ZIKV infection in the target host. Treating mice with anti-LTRIN limited the infectivity and spread of ZIKV in the mouse’s bodies; this points to a potential target for interventions against ZIKV.
Q: What observations can we make on WHY the early immune response to ZIKV is changed when LTRIN is present?
A potential explanation for this effect in LTRIN-ZIKV mice is that the spleens of these mice lacked macrophages and neutrophils. This absence suggests ZIKV gains an advantage, via LTRIN, by suppressing early immune reactions and innate cell types.
In summary, Jin et al showed than LTRIN specifically inhibits a critical axis of communication from skin/epithelial cells to local innate immune cells, enabling ZIKV to spread further and faster in the host.
Q: What’s next?
The findings in this paper have illuminated two potential paths for intervention- targeting the action of LTRIN in the mosquito or the host, and targeting the AXL / NFκB pathway in the host. If, for example, AXL could be selectively suppressed or made unavailable to ZIKV, the virulence and speed of infection might be reduced. Similarly, restoration of the NFκB signaling pathway might boost host defense against ZIKV, resulting in a shorter or less severe disease course.
Learn more about:
Article: Salivary factor LTRIN from Aedes aegypti facilitates the transmission of Zika virus by interfering with the lymphotoxin-β receptor. Lin Jin, Xiaomin Guo, Chuanbin Shen, Xue Hao, Peng Sun, Pengpeng Li, Tao Xu, Chunmiao Hu, Ombati Rose, Hongning Zhou, Mingdong Yang, Cheng-Feng Qin, Jingya Guo, Hua Peng, Mingzhao Zhu, Gong Cheng, Xiaopeng Qi & Ren Lai
Nature Immunology volume 19, pages342–353 (2018)
Featured Image: Fotoshop Tofs @ https://pixabay.com/en/mosquito-female-aedes-albopictus-1332382/